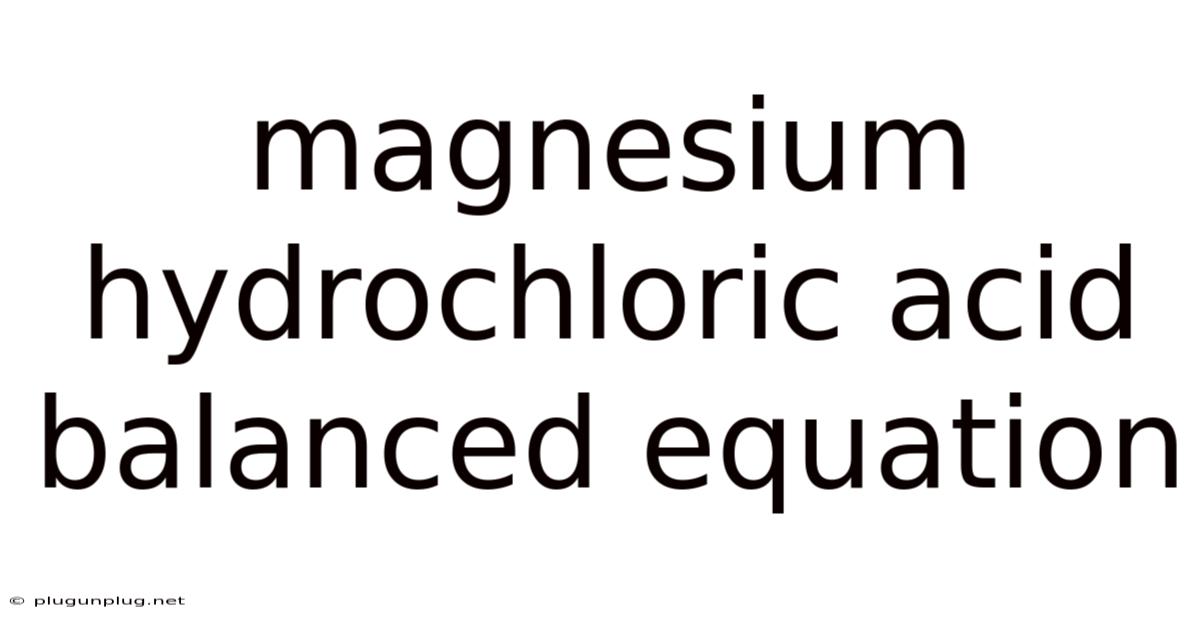Understanding the Reaction Between Magnesium and Hydrochloric Acid: A practical guide
The reaction between magnesium (Mg) and hydrochloric acid (HCl) is a classic example of a single displacement reaction, often used in introductory chemistry courses to illustrate fundamental concepts like reactivity, acid-base reactions, and stoichiometry. In real terms, this article will delve deep into this reaction, exploring its balanced equation, the underlying chemistry, safety precautions, and practical applications. We'll also address frequently asked questions to ensure a comprehensive understanding. Understanding this seemingly simple reaction provides a strong foundation for comprehending more complex chemical processes.
Introduction: The Fundamentals of the Reaction
Magnesium, an alkaline earth metal, is relatively reactive. Now, this reaction is exothermic, meaning it releases heat. But when magnesium comes into contact with hydrochloric acid, a vigorous reaction occurs, producing magnesium chloride (MgCl₂) and hydrogen gas (H₂). Hydrochloric acid, a strong acid, readily donates protons (H⁺ ions). The key to understanding this reaction lies in the balanced chemical equation, which accurately represents the stoichiometry – the quantitative relationship between reactants and products.
You'll probably want to bookmark this section That's the part that actually makes a difference..
The Balanced Chemical Equation: A Precise Representation
The unbalanced equation for the reaction between magnesium and hydrochloric acid is:
Mg(s) + HCl(aq) → MgCl₂(aq) + H₂(g)
This equation shows the reactants (magnesium and hydrochloric acid) and the products (magnesium chloride and hydrogen gas). Even so, it's not balanced because the number of atoms of each element isn't equal on both sides of the equation. To balance the equation, we need to adjust the coefficients (the numbers in front of each chemical formula) to confirm that the number of atoms of each element is the same on both sides:
Mg(s) + 2HCl(aq) → MgCl₂(aq) + H₂(g)
This balanced equation shows that one mole of magnesium reacts with two moles of hydrochloric acid to produce one mole of magnesium chloride and one mole of hydrogen gas. This ratio is crucial for understanding the quantitative aspects of the reaction, such as determining the amount of products formed from a given amount of reactants.
A Step-by-Step Explanation of the Reaction Mechanism
The reaction proceeds through a series of steps involving the transfer of electrons:
-
Protonation: The hydrochloric acid molecule dissociates in water to form hydronium ions (H₃O⁺) and chloride ions (Cl⁻). The hydronium ion acts as a source of protons Nothing fancy..
-
Electron Transfer: Magnesium atoms readily lose two electrons to achieve a stable electron configuration (like that of neon). These electrons are transferred to the hydrogen ions (H⁺) from the hydrochloric acid Not complicated — just consistent..
-
Formation of Magnesium Ions: The magnesium atoms, having lost two electrons, become positively charged magnesium ions (Mg²⁺).
-
Formation of Hydrogen Gas: Each pair of hydrogen ions gains two electrons to form a hydrogen molecule (H₂), which is a gas at room temperature and escapes as bubbles Took long enough..
-
Formation of Magnesium Chloride: The magnesium ions (Mg²⁺) and chloride ions (Cl⁻) are attracted to each other due to electrostatic forces, forming magnesium chloride (MgCl₂), which dissolves in water to form an aqueous solution.
This process can be summarized as an oxidation-reduction (redox) reaction where magnesium is oxidized (loses electrons) and hydrogen is reduced (gains electrons).
Understanding the Role of Hydrochloric Acid Concentration
The concentration of the hydrochloric acid significantly impacts the rate of the reaction. Also, this is because the frequency of collisions between magnesium atoms and H⁺ ions increases with higher concentration, leading to a faster rate of electron transfer and subsequent product formation. Think about it: a higher concentration of HCl means a greater number of H⁺ ions are available to react with magnesium, leading to a faster reaction rate. You will observe a more vigorous reaction, with more rapid bubbling and a higher temperature increase, when using a more concentrated solution of HCl And that's really what it comes down to..
Safety Precautions: Handling Acids Responsibly
Hydrochloric acid is a corrosive and potentially dangerous substance. It's crucial to handle it with appropriate safety precautions:
- Eye Protection: Always wear safety goggles or a face shield to protect your eyes from splashes.
- Gloves: Wear chemical-resistant gloves to prevent skin contact.
- Ventilation: Perform the reaction in a well-ventilated area or under a fume hood to avoid inhaling hydrogen gas or acid fumes.
- Appropriate Clothing: Wear a lab coat or apron to protect your clothing.
- Disposal: Follow proper procedures for disposing of the used acid and reaction by-products. Never pour acids down the drain without proper neutralization.
Hydrogen gas is flammable and can form explosive mixtures with air. check that there are no open flames or sparks near the reaction setup.
Practical Applications: Beyond the Classroom
The reaction between magnesium and hydrochloric acid, while often studied in a classroom setting, has several practical applications:
- Production of Hydrogen Gas: The reaction can be used as a convenient method for producing small amounts of pure hydrogen gas in a laboratory setting. Hydrogen has many applications, including fuel cells and industrial processes.
- Corrosion Prevention: Magnesium is often used as a sacrificial anode in corrosion protection systems. It reacts preferentially with corrosive agents, protecting more valuable metals from degradation. Understanding the reaction with acids helps in designing and optimizing these protection systems.
- Determination of Magnesium Content: The reaction can be used in analytical chemistry to determine the amount of magnesium present in a sample through quantitative analysis. By measuring the amount of hydrogen gas produced, the initial amount of magnesium can be calculated using stoichiometric relationships.
- Educational Tool: As noted, this reaction is a fundamental concept in introductory chemistry, illustrating several important principles and allowing students to develop skills in balancing equations, performing calculations, and understanding reaction mechanisms.
Frequently Asked Questions (FAQs)
Q: What are the observable changes during the reaction?
A: You'll observe vigorous bubbling due to the formation of hydrogen gas. The magnesium strip will gradually dissolve, and the solution will become warmer due to the exothermic nature of the reaction.
Q: Can other acids be used instead of hydrochloric acid?
A: Yes, other strong acids like sulfuric acid (H₂SO₄) and nitric acid (HNO₃) will also react with magnesium. That said, the reaction rate and products might differ slightly. Nitric acid, in particular, is a strong oxidizing agent and can lead to the formation of different products And that's really what it comes down to..
Q: What happens if the magnesium is impure?
A: Impurities in the magnesium may affect the reaction rate and potentially introduce other reaction by-products. Pure magnesium is preferred for accurate experimental results.
Q: How can I determine the rate of the reaction?
A: The rate can be determined by measuring the volume of hydrogen gas produced over time. This can be done using a gas collection apparatus and measuring the gas volume using a graduated cylinder or other volumetric equipment Practical, not theoretical..
Q: What are the hazards associated with hydrogen gas?
A: Hydrogen gas is flammable and can form explosive mixtures with air, particularly at certain concentrations. It's crucial to avoid open flames and sparks when working with hydrogen gas. Good ventilation is essential to prevent the accumulation of hydrogen gas in enclosed spaces Not complicated — just consistent..
Conclusion: A Foundational Reaction with Broad Implications
The reaction between magnesium and hydrochloric acid, while seemingly simple, provides a rich learning experience, illustrating fundamental concepts in chemistry and demonstrating practical applications across various fields. Understanding the balanced equation, the reaction mechanism, and the associated safety precautions is essential for both students and professionals working with these chemicals. In practice, this reaction serves as a stepping stone to understanding more complex chemical systems and reactions. Here's the thing — the careful study of this seemingly simple reaction allows us to gain insights into the intricacies of chemical interactions and their importance in the world around us. Remember always to prioritize safety when conducting any chemical experiment The details matter here..